Types of IVD Devices
-
 +
+
- Smart Blood Glucose Measurement Tester HZ Blood Glucose Meter Home BHM-102
- Mircotouch Blood Sugar Glucose Meter Glucometer Monitoring System BGM-102
- HZ CE Approved Household Usage Quick Test Sugar Test Kit
- Customization Acceptable Handheld Electronic Home Use Blood Glucose Meter BGM-101/101N
- Code Free Diabetes Test Meter Glucose Monitor BGM-102
- Portable Blood Sugar Test Meter BGM-102
- HZ GULP-101 Triglycerides And Heart Rate Checker Blood Glucose Tester For Home Use
- Handheld Customizable Electronic Home Use Glucometer Blood Glucose Meter BGM-102
- Customize Blood Glucose Meter Blood Sugar Glucometer Monitor Diabetes Horse Dog Cat BGM-102
- High Precision Blood Glucose Meter Diabetes Monitor BGM-102
- Simple Operation Smart Glucose Monitor Diabetes Detecting
- HZ Diabetes Digital Glucometro Blood Gluco Meter, Blood Sugar Monitor Kit Blood Glucose Testing Machine
- Mini Blood Glucometer 5-second Accurate Result Diabetes Glucose Monitor Home Blood Glucose Meter CE ISO Apprpved
- Direct Sale Blood Glucose Meter For Household BGM101/101N
- HZ EGS-101 Blood Sugar Test Strips For Intelligent Glucose Analysis Instrument
- HZ GUM-101 Blood Glucose/Uric Acid Tester
- ISO9001 Fresh Capillary Blood Glucose Testing With HZ GUM-101
- 200 Records High Accuracy Glucose Meter With Strips BGM101
- HZ BGM-102 Blood Sugar Test Meter LCD Display Glucometer Kit
- HZ Glucometer Blood Sugar Test Diabetes Check Machine 50g
- HZ Diabetes Test Kit Portable Glucose Monitoring Devices BGM-102
- Electrochemistry Method HZ Glucometer Test Strips Sugar Monitor For Diabetes
- BGM-101 Diabetic Test Strips Blood Glucose Monitor
- Microinvasive Portable Blood Glucose Monitor Medical Device BGM-101
- Fresh Capillary Portable Blood Glucose Meter Sugar Check Machine BGM-102
- Large LCD Diabetes Test Meter Portable Blood Glucose Meter
- HZ GUM-101 Uric Acid Test Strips Portable Blood Sugar Tester 50g
- RoHS HZ Blood Glucose Tester GUM-101 For Hassle-Free Testing
- Smart HZ GULP-101 Blood Glucose Tester Multi-Function Glucose Analyzer
- 365g Blood Glucose Tester HZ GULP-101 Cholesterol And HDL Analyzer
- HZ GUM-101 Smart Data Analysis Blood Glucose Tester Uses Electrochemistry Method
- HZ GUM-101 Blood Glucose Tester 7*52*18mm Uric Acid Test At Home
- Glu 50 Records HZ GUM-101 Blood Glucose Tester Strips 50g
- HZ GUM-101 3.0V CR2032 Blood Glucose Tester Strip Uric Acid Testing
- UA 50 Records Uric Acid Blood Glucose Tester With HZ GUM-101
-
 +
+
- Reflectance Photometer Hemoglobin Tester HCT Testing With HZ BHM-101
- HZ BHM-101 Hemoglobin Tester Clinical Use Hematocrit Analyzer
- RoHS Reflectance Photometer Hemoglobin Tester With HZ BHM-101 Machine
- 1200mAh Hemoglobin Tester HCT Analyzer With User-Friendly Interface BHM-101
- High Precision BHM-101 Hemoglobin Tester HCT Analyzer Test For Medical Use
- CE Hemoglobin Tester Hemoglobin And Hematocrit Machine 800 Records
- BHM-101 HCT Analyzer Hemoglobin Tester For Accurate Blood Analysis Test
- 800 Records Hemoglobin Tester Hemoglobin HCT With HZ BHM-101 Analyzer
- Fast And Accurate 90% RH Hemoglobin Tester HCT Analysis With BHM-101 By HZ
- HZ Blood Hemoglobin HCT Analysis Meter For Home / Hospital Use BHM-102
- 800 Records Glycosylated Hemoglobin Test Hba1c Test Fasting With HZ BHM-102
- BHM-102 Hemoglobin HCT Analyzer Accurate Blood Glucose Meter
- Precise Hemoglobin HCT Measurement With HZ BHM-102 Hematology Analyzer
- User-Friendly Hemoglobin HCT Test Analyzer With BHM-102 By HZ
- ISO9001 Hba1c Diabetes Hemoglobin Blood Test With HZ BHM-102
- Hgb Blood Test Hemoglobin HCT Analyzer By HZ BHM-102 Test
- 1200mAh Microtouch Home Hemoglobin Analysis Digital Meter BHM-101
- Digital Portable Noninvasive Blood Hemoglobin Meter HZ BHM-102
- 800 Records Home Hemoglobin Test Meter Machine Portable BHM-102
- Precise Hemoglobin Tester HCT Measurement With BHM-101 Analyzer
- Electronic Digital HB Meter Hemoglobin Analyzer
- Hemoglobin Analyzer HB Test Meter
- Hospital Or Family Use Portable Hemoglobin Meter
- Rapid Self Test Blood Hemoglobin Analyzer Meter BHM-101
- Fully Automatic Hemoglobin Meter Analyzer
- Portable Handheld Hemoglobin Analysis Meter
- Hemoglobin Test Paper
-
 +
+
- Uric Acid Testing Kits Home Uric Acid Blood Level Tester Machine GUM-101
- GUM-101 Home Blood Uric Acid Self Test Kit
- Rapid Diabetes Test Uric Acid Testing Kit With GUM-101
- GUM-101 Rapid Blood Glucose Test 50g Glu 50records UA 50 Records
- 1min Automatic Shut Off GUM-101 Cholesterol Glucose Uric Acid Test 50g
- Intelligent Recognition GUM-101 Automatically Uric Acid Meter Test Kit
- Heart Rate Blood Pressure Monitor Uric Acid Test Meter GULP-101
- GULP-101 Multi-Parameter Analyzer Uric Acid Test Machine 365g
- GULP-101 HDL Glucose Cholesterol Uric Acid Meter For Diabetes Management Uric Acid Test
- Compact And Lightweight GUM-101 Portable Uric Acid Test Meter
- RFM-101 Rechargeable Uric Acid Tester With Bluetooth Connectivity
- Home Uric Acid Tester With RFM-101 Reflectance Photometer
- Monitoring Uric Acid Levels Uric Acid Tester With RFM-101 Portable Meter
- 500 Records Gout Test Kit Uric Acid Tester With RFM-101 Reflectance Photometer
- CE Uric Acid Tester Bluetooth Enabled Blood Glucose Meter 90g
- Streamline Uric Acid Tester Bluetooth Enabled Glucose Meter RFM-101
- RoHS Uric Acid Tester With RFM-101 Reflectance Photometer And Mobile App
- 1200mAh Uric Acid Tester With RFM-101 Rapid Test Meter
- RFM-101 Reflectance Photometer Uric Acid Tester Solution
- 500 Records Uric Acid Tester With RFM-101 Portable Reflectance Photometer
- Easy Touch Uric Acid Test Kit
- Uasure Uric Acid Test Strips
- Blood Glucose Uric Acid Meter Multi-function Glucometer Uric Acid Meter
- Kidney Function Test Strips
- Home Use Multifunction Monitoring System Glucose Uric Acid Analyzer
- 2 in 1 Machine High Precision Uric Acid Blood Glucose Analyzer
- 2 in 1 Blood Glucose & Uric Acid Meter Tiny Specimen Health Care
- High-precision Uric Acid Meter
- Glucose Uric Acid Analysis Meter
- Blood Glucose Meter Uric Acid Monitor With Test Strip
- Intelligent Model Multifunctional Analysis Meter Glucose Uric Acid
- Auto Recognize Strips Uric Acid Test Strip
- Diabetes Uric Acid Tester
- Electrochemistry Method 2 in 1 Glucose& UA Multifunction Analysis Meter
- Digital Sugar Uric Acid Test Strip Active Blood Glucose Meter
-
 +
+
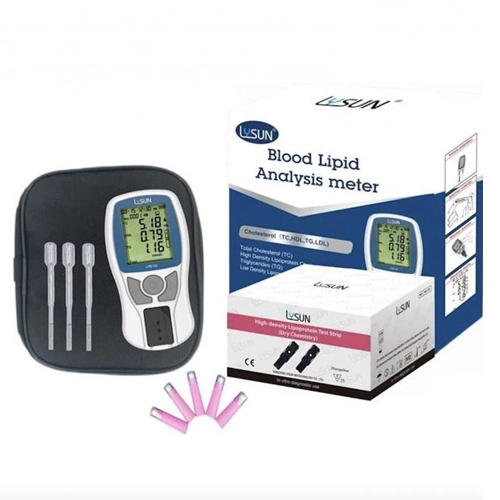
- Blood Lipid Analyzer PFS-30 (With Bluetooth) PFS-30A
- 4 in 1 Panel Quick Blood Lipid Analyzer For TC, TG, HDL, LDL
- LPM-102 Lipid Profile Panel Blood Test For TC / HDL / TG / LDL Analysis
- Lipid Profile Test Kit Analyzer LPM-102 For Cardiovascular Risk Assessment
- 500 Records Blood Lipid Analysis Meter Portable Lipid Analyzer 90g
- LPM-102 Lipid Profile Machine For LDL Analysis With Intelligent Printer Linkage
- LPM-102 Lipid Profile Test Machine For Comprehensive Lipid Analysis
- LPM-102 Lipids Blood Test Machine With TC/HDL Ratio Calculation
- LPM-102 Lipid Profile Test 500 Records Portable Lipid Test System
- LPM-102 Lipid Test Device Health Monitoring Triglycerides Tester
- Multiple Functions 500 Records Lipid Meter LPM-102
- LPM-102 Blood Lipid Test Meter 500 Records
- DBM-101 Renal Function Test Lipid Tester 3 In 1 Cholesterol Meter
- DBM-101 Fast Blood Lipid Tester With Bluetooth Connectivity
- DBM-101 Rechargeable Blood Lipid Tester With Long Battery Life
- DBM-101 Digital Blood Lipid Tester With Large Memory And Auto Shut-Off
- Portable DBM-101 Comprehensive Blood Lipid Tester Easy-To-Read Display
- Comprehensive Blood Lipid Tester DBM-101 With HDL TG And TC Analysis
- Health Monitoring DBM-101 Lipid Tester Renal Function Testing
- DBM-101 TC HDL TG Measurements Lipid Tester 500 Records
- DBM-101 Portable Blood Lipid Tester Renal Function Test Solution
- DBM-101 Lipid Tester Renal Function Testing With Bluetooth Syncing
- Triglycerides Test Strip
- Triple-in-one Lipid Monitor HDL LDL Triglycerides Cholesterol
- Multi Parameters Dry Biochemistry Analyzer For Blood Lipid, Renal Function
- Handheld Cholesterol Blood Lipid Meter Monitoring
- 3 In 1 Blood Lipid Test Strip Total Cholesterol, Triglycerides, High Density Lipo-protein TC/TG/HDL/LDL For POCT Analyzer
- IVD Reagent Lipid Profile Dry Chemistry Test Strips By Meter Portable Lipid Profile Analyzer
- Dry Chemistry Analyzer Blood Lipid Renal Function Analysis Meter
- Medical Home Used Portable Blood Lipid Meter
- Total Cholesterol HDL Triglycerides LDL Blood Lipid Analyzer Meter For Hospital Diagnostic Agency
- Medical Equipment Total Cholesterol Creatinine Meter Dry Biochemical Analyzer Renal Function Analysis Meter
- CE 6 in 1 TC/TG/HDL/UR/UA/CR Dry Biochemical Analysis Meter For Cholesterol
- Household Medical Cholesterol Meter
-
 +
+
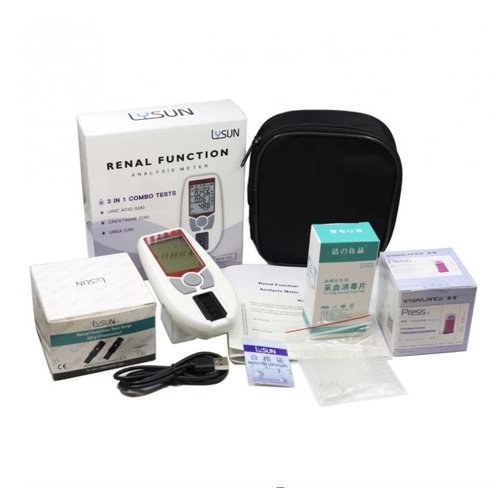
- 5 Mins Blood Test Machine Renal Kidney Profile Test RFM-101
- Dry Biochemical Analysis Creatinine Test Strip Renal Function Analysis Meter
- DBM-101 Portable Blood Lipid And Renal Function Panel Test 90g
- DBM-101 Kidney Disease Blood Test Bluetooth Enabled
- DBM-101 Multi-Functional Blood Lipid And Renal Function Blood Test
- DBM-101 Kidney Blood Test Blood Lipid Analyzer 500 Records
- DBM-101 Blood Lipid Analysis Meter Test For Kidney Function
- DBM-101 Blood Lipid Renal Function Analysis Meter 500 Records
- Kidney Function Test Dry Biochemical Analysis Meter DBM-101
- RFM-101 Rechargeable Uric Acid Test Meter With Bluetooth Connectivity
- Renal Function Testing With DBM-101 UA CR And UR Analysis
- DBM-101 Precise Blood Lipid And Renal Function Test Meter For Home Use
- RFM-101 Renal Function Test With Rapid Results And Bluetooth Connectivity
- 300 Second Rapid Testing Renal Function Test FM-101 Automatically Update
- Kidney Health RFM-101 Bluetooth-Enabled Renal Function Test 90g
- RFM-101 Rechargeable Renal Function Test Results In 300 Seconds
- Renal Function Testing With RFM-101 Bluetooth Syncing And Mobile App Management
- 500 Records Renal Function Testing With RFM-101 Whole Blood Plasma And Serum Specimen Capability
- RFM-101 Kidney Health Monitoring Renal Function Test With 1200mAh Li-Ion Battery
- RFM-101 Renal Function Test 500 Records With User-Friendly Mobile App
- 3 in 1 Renal Function Test Strip
- Urea Analysis Meter
- Renal Function Analysis Meter
- Kidney Function Test Strip
- Clinical Analytical Renal Function Analyzer Creatinine Test
- Economic Renal Function Analysis Meter
- Long Endurance Renal Function Meter
- Safe-accu Renal Function Test Machine For Creatinine, Uric Acid, Urea
- Economic Home Renal Function Analysis Digital Meter
- New Design Renal Function Analyzer For Uric Acid(UA)/Creatinine(CR)/Urea(UR) Clinic Home Use
- Health Care Digital Renal Function Analysis Digital Meter And Strips
- Automatic Chemistry Analyzer Portable CE Biochemistry Analyzer For Liver Renal Function Test
- Handheld Renal Function Analyais Meter
- Renal Function UR CR UA Test Meter
- Renal Function Analysis Meter And Strips
- Dry Biochemical Analysis Creatinine Meter Medical Health Care Blood Lipid Renal Function Analysis Meter
-
 +
+
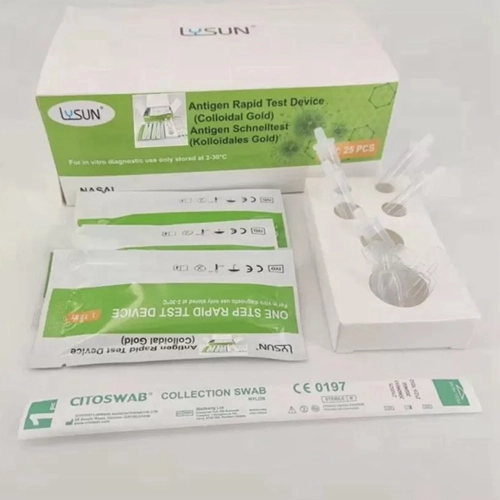
- Rapid and Accurate HCV Antibody Testing with HCVAb Cassette Test HCV-W11
- MP Biomedicals Rapid Antigen Test For Pneumonia Diagnosis MP-P21-GM
- Flu A+B Test With Strip Format Antigen Test Infectious FLU-S11
- Diagnosis Of Syphilis With SYP Rapid Test Cassette SYP-W21
- Malaria Test Diagnosis With MAL-W21-FA Rapid Test Cassette
- Torch Rapid Test Kit Simultaneous Detection Of Rubella CMV HSV And TOXO Antibodies TOR-W24-GM
- Antigen Test Infectiousness Diagnosis With CHA Test For Chagas Disease CHA-101
- TOXO IgM Test For Early Detection Of Toxoplasmosis TOX-W21-M
- FLUA+B COVID-19 RSV Rapid Test For Respiratory Infections
- HBcAb Test Cassette For Hepatitis B Virus Detection HBcAb-W21
- Hepatitis B E Antigen Detection With HBcAg Test Cassette HBcAg-W21
- Swab Strep A Testing Strip Antigen Test Infectious STP-S11
- HP-P21 Antigen Test Infectious H Pylori Antigen Rapid Test Cassette
- Helicobacter Pylori Test Antigen Test Infectious HP-W11
- Strep A Rapid Test Cassette Antigen Test Infectious STP-S21
- Syphilis Detection With SYP Test Strip Antigen Test Infectious SYP-P11
- Syphilis Pregnancy Test Strip Antigen Test Infectious SYP-W11
- Rapid And Accurate Malaria Testing With MAL-W21-FV Cassette Antigen Test Infectious
- HIV HBsAg Rapid Antigen Test Infectious BIV-W22 Cassette
- EV71 IgM Rapid Test Antigen Test Infectious EV-W11 Cassette
- Fast and Accurate Chikungunya Virus Detection with CHI Rapid Test
- Rapidly Detect HSV IgG/IgM with Our HSV Test Cassette HSV-W21-GM
- Fast and Accurate HSV IgM Testing with the HSV Rapid Test HSV-W21-M
- Fast and Accurate Diagnosis of Rubella with Rubella IgG/IgM Rapid Test RUB-W21-GM
- Fast and Accurate Diagnosis with Rubella IgM Rapid Test Kit RUB-W21-M
- Fast and Accurate CMV IgG/IgM Testing with CMV Rapid Test CMV-W21-GM
- Fast and Accurate CMV IgM Rapid Test for the Detection of Cytomegalovirus Infections CMV-W21-M
- Quick and Accurate TOXO IgG/IgM Testing with Our Rapid Test Kit TOX-W21-GM
- Rapid Torch Test for Accurate Detection of Rubella, CMV, HSV, and TOXO IgM Antibodies TOR-W24-G
- Fast and Accurate Detection of Multiple Infections with HBsAg/HCV/HIV/Syphilis Combo Test BCIS-P24
- Fast and Accurate Diagnosis with HBsAg/HCV Rapid Test Cassette BCV-W22
- Rapid and Accurate Diagnosis of Malaria with MAL-W11-FA Strip Test
- Rapid and Accurate Detection of Malaria Pf/Pv with the Malaria Test Strip MAL-W11-FV
- Quick and Accurate Malaria Pf Detection with our Rapid Test Strip MAL-W11-F
- Fast and Accurate H. Pylori Testing with the HP-F21 Rapid Test Cassette
- Rapid H. Pylori Test Strip for Accurate Diagnosis Antigen Test Infectious HP-P11
- Rapid Test for Detection of Influenza A and B Antigen Test Infectious FLU-S21
- Rapid FLU B Test for Quick and Accurate Diagnosis Antigen Test Infectious IFU-S21
- Rapidly Detect HAV Infections with HAV Ag Test Cassette HAV-W31-AG
- Infectious Disease Rapid Test: MP IgM Test for Rapid and Accurate Diagnosis MP-P11
- Rapid HAV IgM Testing with HAV Test Cassette for Early Detection HAV-P11
- Rapid and Accurate Testing for Hepatitis B, Hepatitis C, HIV, and Syphilis with BCIS Test BCIS-W11
- Fast and Accurate Diagnosis of Cholera with CHO Test Cassette CHO-F21
- Fast and Accurate Diagnosis of Cholera with CHO Test Cassette Antigen Test Infectious CHO-F11
- Rapid and Accurate Rotavirus Testing with ROTA, ADENO Test Cassette ROT-F21
- Rapid and Accurate Typhoid/Para Typhoid Ag Detection with TPPA-F22 Test
- Fast and Accurate Typhoid Ag Test for Fecal Specimens Antigen Test Infectious TYP-F21
- Rapid and Accurate Typhoid Test with Typhoid IgG/IgM Cassette TYP-W21
- Fast and Accurate Typhoid Test for Early Diagnosis Antigen Test Infectious TYP-P21
- Fast and Accurate HCV Antibody Testing with HCVAb Cassette Test Antigen Test Infectious HCV-W21
-
 +
+
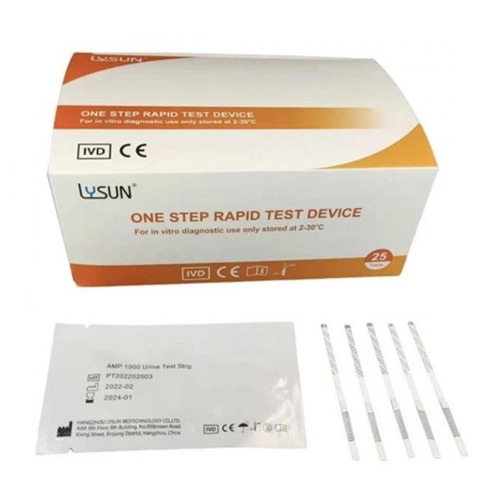
- Quick and Accurate Detection of PPX in Urine Using Rapid Diagnostic Cassette PPX-U102
- Accurately Detect EDDP in Urine with Our Rapid Diagnostic Drug Test EDDP-U102
- Rapid Diagnostic Drug Test Cassette For MET Detection In Urine MET-U102
- 200ng/ML Detection Of TRA Rapid Diagnostic Test Cassette TRA-U102
- 30ng/ML 50ng/ML K2 Urine Drug Test Diagnostic Drug Test Cassette
- Fast Detection Of Cotinine COT Medical Drug Urine Test 200ng/ML
- Urine ETG-U103 Drug Of Abuse Test ETG Test Kit Panel
- Rapid Diagnostic Drug Test Strip For MET Detection In Urine Drug Of Abuse Test MET-U101
- Rapid Detection TRA Drug Test Drug Of Abuse Test 200ng/ML
- 100ng/ML TML Urine Drug Test Strip Drug Of Abuse Test TML-U101
- Rapid Diagnostic For Saliva Samples Drug Of Abuse Test ACO-U101
- ETG Testing For Alcohol Consumption Drug Of Abuse Test ETG-U101
- Drug Of Abuse Test ETG Test Cassette For Alcohol Detection In Urine ETG-U102
-
 +
+
- Pregnancy Detection Fetal Fibronectin Test HCG Fertility Test 25mIU/Ml
- Healthcare Serum Urine HCG Pregnancy Test Cassette 25mIU/Ml
- CE HCG Midstream Fertility Test One Step HCG Urine Test 25mIU/ml
- 25mIU/Ml HCG Pregnancy Test Strip Kit In Urine For Serum
- Rapid HCG Test Cassette For Serum Detect Pregnancy Early And Accurately HCG-P21
- 25mIU/ml HCG Test For Fertility Monitoring HCG-M21 Fertility Test
- LH Test Strip For Accurate Ovulation Prediction Fertility Test LH-U11
- 25mIU/Ml LH Ovulation Rapid Test Cassette Detect Luteinizing Hormone
- 25mIU/Ml Fertility Monitoring LH Test Strip Fertility Tester LH-U31
- FSH Urine Test Cassette For Fertility Evaluation Fertility Tester FSH-U21
- 25mIU/Ml Midstream FSH Urine Test Fertility Tester Detects FSH Levels
- IGFBP-1 Urine Test For Fertility Assessment FBP-U11 Fertility Tester 50pcs
- Rapid FFN Test Strip Fertility Tester 25ng/Ml Diagnose The Risk Of Preterm Labor
- Fertility Tester With FFN Urine Test Cassette FFN-U21
-
 +
+
- High Sensitivity Cardiac Risks CRP Test For Early Detection Cardiac Marker Test CRP-101
- Cardiac Diseases Early Detection CTI Test Cardiac Marker Test 0.3ng/Ml 0.5ng/Ml
- Detect Cardiac Ischemia CTI Test Cassette Cardiac Marker Test CTI-P21
- 5ng/Ml CKMB Test For Rapid Cardiac Marker Test Detection Cassette CKMB-W21
- Myoglobin Detection MYO Test MYO-W21 Cardiac Marker Test 50ng/Ml
- PCT Testing With PCT Test Strip PCT-W11-1 Cardiac Marker Test 0.5ng/Ml
- PCT-W21-1 PCT Test Cassette Cardiac Marker Test 0.5-10 Ng/ML
- Reliable and Fast Results with the SAA Test Strip and Cassette SAA-W21
- Rapid SAA Test for Early Detection of Systemic Inflammation SAA-W11
- High-Sensitivity CRP Test Cassette for Efficient Detection of Cardiovascular Diseases CRP-W21-2
- Detect Inflammation and Monitor Disease Progression with CRP Test Strip CRP-W21-1
- Accurately Detect Procalcitonin Levels with the PCT Test Cassette PCT-W21-2
- High Sensitivity PCT Test Strip for Rapid Detection of Infection PCT-W11-2
- Rapid CRP Test for Quick and Accurate Diagnosis of Inflammatory Conditions CRP-102