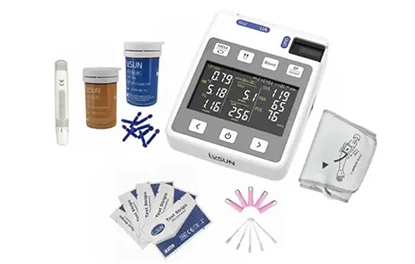
Understanding the Technology and Applications of In Vitro Diagnostic Devices
What is In Vitro Diagnostic Device?
In Vitro Diagnostic (IVD) instruments detect human samples (various body fluids, cells, tissue samples, etc.) to obtain clinical diagnostic information externally, thereby determining diseases or bodily functions. Known as the "eyes of doctors" in the medical field, in vitro diagnostic instruments are a crucial part of modern clinical laboratory medicine. The clinical application of IVD devices spans the entire process of disease diagnosis and treatment, including disease prevention, initial diagnosis, treatment plan selection, and efficacy evaluation, providing doctors with a large amount of useful clinical diagnostic information, becoming an indispensable part of human disease diagnosis and treatment.
The design and development of in vitro diagnostic instruments, as specialized devices, are based on medical laboratory science and in vitro diagnostic methodologies. With the development of in vitro diagnostic methodologies, detection-related technologies, instrumentation engineering, etc., the design of in vitro diagnostic devices is increasingly refined, gradually reaching advanced levels of high-end medical equipment development. In vitro diagnostics can be categorized into biochemical diagnostics, immunodiagnostics, molecular diagnostics, microbiological diagnostics, hematological diagnostics, and Point-of-Care Testing (POCT). The related technologies include electrochemical technology, spectrometry, chromatography, mass spectrometry, electrophoresis, flow cytometry, labeled immunoassay technologies, molecular biology, and biosensor technology. The in vitro diagnostic instruments are diverse, and their development process generally includes stages such as product project design, product prototype development, product finalization, and product acceptance, with each stage being based on certain rules.
Related Technologies and Applications of In Vitro Diagnostic Devices
Although there are many types of in vitro diagnostic instruments used in clinical applications, their design and development are always based on the emergence and development of related technologies. In vitro diagnostic device is grounded in clinical and laboratory methodologies, integrating automation technology, optical technology, electronic information technology, biosensor technology, computer technology, among others, progressively evolving into systematic, engineered modern instruments and equipment to meet the needs of clinical trials and medical services.
Electrochemical Analysis Technology: Electrochemical analysis involves forming a chemical cell from a solution of the substance to be analyzed and converting the concentration of the substance into electrical parameters by measuring variables like potential, current, power, and resistance. Electrochemical analysis can be divided into potentiometric analysis, conductometry, electrolysis, capacitive analysis, voltammetry, and electrochemical biosensor unit technology. Most instruments for electrolyte analysis, blood gas and pH analysis in clinical biochemistry testing are based on electrochemical analysis technology. Electrochemiluminescence Immunoassay (ECLIA) combines the chemiluminescence process induced by electrochemical reactions with immunoreactions. ECLIA has high sensitivity of luminescence detection and high specificity of immunoassay. Electrochemiluminescence automated immunoassay instruments based on ECLIA technology are widely used in clinical immunology tests, including the determination of tumor markers, hormones, enzymes, antigens or antibodies, vitamins, cytokines, and various metabolites.
Spectroscopy Technology: Spectroscopy involves the absorption, emission, or scattering of light of different wavelengths and intensities when it interacts with matter to characterize and quantitatively analyze a substance. According to the method of generating the spectrum, spectroscopy can be classified into three main types: absorption spectroscopy, emission spectroscopy, and scattering spectroscopy. In vitro diagnostic instruments using spectroscopy are relatively simple, easy to operate, and methodically sensitive, thus widely used in clinical testing. Absorption spectra, which are obtained from the selective absorption of radiation energy by molecules or atoms, include ultraviolet-visible spectrophotometry, infrared spectroscopy, and atomic absorption spectrophotometry. Ultraviolet-visible spectrophotometry is mainly used for quantitative checks of substances, such as serum inorganic phosphate. Atomic absorption spectrophotometry is mainly for the analysis of metal elements like zinc, magnesium, and copper in the blood. Emission spectra are produced when molecules, atoms, or ions of a substance absorb external energy and transition from the ground state to a high-energy state and then return to the ground state. Emission spectroscopy involves characterizing substances through quantitative checks using emission spectra. Fluorescence spectrophotometry is more commonly used in clinical testing. For example, it can be used to determine vitamin content. When photons interact with a heterogeneous dielectric, light scattering occurs; measuring the intensity of scattered light provides the content of the analyte. As light passes through the sample, the antigen forms antigen-antibody complexes with its specific antibody, increasing solute particles and scattering intensity proportional to the complex concentration. This scattering intensity change is used to measure antigen content. Automated scattering turbidimetry analyzers, based on this scattering turbidimetry, are widely used in clinics, mainly for measuring protein substances like serum immunoglobulin, C-reactive protein, and trace proteins in urine.
Chromatography Technology: Chromatography is a collective term for separation and analysis techniques, mainly used for the separation and analysis of complex multicomponent mixtures. It involves using differing partition coefficients between immiscible phases to separate components of a mixture, then characterizing or quantitatively determining them. Chromatography includes many types and can be divided by mobile phase characteristics into liquid chromatography and gas chromatography; by separation principles into adsorption chromatography, partition chromatography, ion-exchange chromatography, etc.; and by the state of the stationary phase into column chromatography and planar chromatography. Column chromatography technology is commonly used in clinical testing.
Mass Spectrometry Technology: Mass spectrometry (MS) generates diagrams of charged atoms, molecules, or molecular fragments arranged by mass-to-charge ratio. Mass spectrometers dissociate substances into ions and use appropriate electric or magnetic fields to separate them based on spatial position, temporal sequence, or orbital stability, analyzing substances after inspecting intensity. Although mass spectrometry can perform qualitative analysis, it cannot analyze mixtures, whereas chromatography effectively separates mixtures; combining them enables simultaneous separation and identification, creating mass spectrometry coupling technology. Current coupling methods include gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS), among others. MS technology is widely used in clinical medicine due to its high sensitivity, low sample consumption, fast analysis speed, and simultaneous integration of separation and identification. For conditions such as bone metabolism disorders, diabetes, cardiovascular diseases, and tumors, LC-MS technology can simultaneously measure 25-(OH) vitamin D2 and 25-(OH) vitamin D3 with high specificity and is considered the gold standard for detecting 25-(OH) vitamin D, becoming increasingly common in clinical applications. Additionally, MS technology is widely used in drug and stimulant testing.
Electrophoresis Technology: Electrophoresis is the movement of charged particles dispersed in a medium toward the anode or cathode under an electric field. Electrophoresis technology, utilizing this phenomenon, primarily separates, identifies, and quantitatively checks proteins and nucleic acids. Common electrophoresis systems are used for analyzing serum proteins, hemoglobin, and isoenzymes.
Flow Cytometry Technology: Flow cytometry (FCM) combines laser technology, fluid dynamics, computing, and electronic measurement, enabling quick and accurate cell measurement in motion. FCM can measure various cell properties, including size, shape, nuclear-to-cytoplasm ratio, pigment content, and cytoplasm granules. It also measures nucleic acid content, chromatin structure, cell surface antigens, or saccharides, and cytoskeletal structures when fluorescence dyes are used. FCM provides high sensitivity, resolution, sorting purity, reproducibility, and multi-parameter information integration, enabling rapid, accurate, and quantitative cell analysis and sorting. Clinically, FCM is primarily used to measure cell surface antigens, such as lymphocyte subsets, hematopoietic stem cells, and antigen-presenting cells, in immunoassays.
Labeled Immunoassay Technology: Labeled immunoassay technology forms the basis for various clinical immunological tests. It typically involves labeling antigens or antibodies with different substances to make them traceable while maintaining antigen-antibody specificity, facilitating the detection of target substances in clinical samples. The primary labeled immunoassay technologies used in clinical immunology include fluorescence immunoassay, radioimmunoassay, enzyme immunoassay, chemiluminescent immunoassay, and electrochemiluminescent immunoassay. Common labeling substances include fluorescein, radioactive isotopes, enzyme proteins, chemiluminescent agents, and electrochemiluminescent agents.
Molecular Biology Analysis Technology: Molecular biology analysis has become a key technique for detecting genetic material of various pathogens, genes causing hereditary diseases, and genes and metabolites related to certain diseases in clinical testing. Polymerase Chain Reaction (PCR) is the most commonly used molecular biology technology for gene amplification. Quantitative Fluorescent PCR technology is widely used for quantifying target molecules, such as quantitating hepatitis B and C viral nucleic acids, crucial for diagnosing diseases and monitoring drug efficacy.
Classification of In Vitro Diagnostic Devices
In vitro diagnostic products mainly consist of diagnostic equipment and diagnostic reagents. In vitro diagnostic instruments fall under clinical testing analytical instruments. The types of in vitro diagnostic devices are diverse, covering almost all medical testing projects.